A recent post at the Scientific America Blog network entitled “‘Chemical’ Is Not a Bad Word” got me to thinking.
My first question was “Should “Is” be capitalized in a title?” But as I’m neither a copy editor, nor a English Language major, I figured that’s a question for greater minds than myself.
No, the real question was, what’s the difference?
As the SA blog says, we are all chemicals and we eat chemicals and use other chemicals to change chemicals into chemicals and then we excrete chemicals. We breathe chemicals. etc.etc.etc.
I did some research. I choose something near and dear to my heart (and because I knew it would generate an interesting result). Vanilla.
There’s natural vanilla flavoring and there’s synthetic vanilla flavoring. Is there a difference? Well, natural vanilla is exceedingly expensive. Up to about $7 or $8 dollars (US) per gram compared to $0.66 (US) per ounce for synthetic vanilla extract. Natural vanilla also has a few things that aren’t in synthetic vanilla, but theses are very, very minor quantities. By far, the most important ingredient is vanillin. That’s what gives the flavor and the smell to vanilla extract.
Natural vanillin starts as this chemical in the green seed pods.
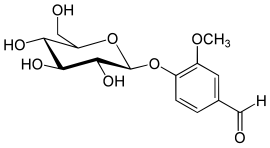 β-D-glucoside
β-D-glucoside
The vanillin is pretty much that part on the right. So it has to be extracted. At this point, it doesn’t smell or taste anything like vanilla. After some super secret processes that involve leaving the green beans in the sun for a couple of weeks, the beans begin to smell like vanilla. The β-D-glucoside has broken down into vanillin.
 vanillin
vanillin
This takes time and any vanilla beans not grown in Central America are hand pollinated (labor costs) and is thus quite expensive. However, there is another way (actually several). (Techniques from here.)
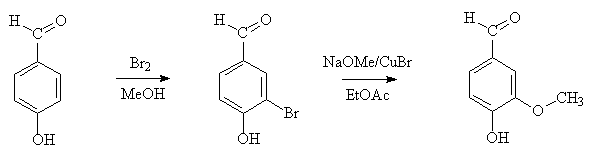
An easy method is to take 4-hydroxybenzaldehyde use a technique of electrophilic bromination and then copper-catalysed methoxylation to produce vanillin.
Or you could start with ‘lesser’ spices like cinnamon and clove (shudder) and put in an alkaline solution. This changes the shape of the compound eugenol to isoeugenol. Then you oxidize this using nitrobenzene into vanillin. And don’t worry about ‘nitrobenzene’, none of that gets into the flavoring. Indeed, nitrobenzene is used in soaps.
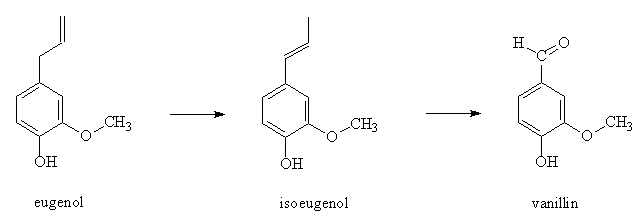
There are at least two other methods to produce vanillin.
OMG, but they aren’t NATURAL methods!1!!!!!!11
So?
Our body doesn’t care where the vanillin came from. All that vanillin does is cause certain chemical reactions to occur when the molecule hits our tongue and nose. What causes those reactions? The shape and composition of the molecule.
Look closely at the three examples I provided. Which version of vanillin is different?
None of them. They are exactly the same. In turn, they will each cause exactly the same reaction in our bodies, which we will perceive as a flavor or smell.
No one could tell the difference between vanilla bean vanillin and vanillin from eugenol by taste or smell. It is not possible. There is a way to tell, but it involves a mass spectrometer and the synthetic vanillin manufacturers have responded by matching vanilla bean vanillin so well, that they even match at an isotope level.
Vanilla extract, however, is a slightly different story. Because the vanilla beans do contain a small, but measurable amount, of other compounds, it may taste different than ‘pure vanilla extract’ which is made without vanilla beans. But my point remains, vanillin is vanillin. It doesn’t matter if it was made from a bean or a petrochemical. It’s exactly the same thing.
Which leads me to wonder, do these people talking about ‘chemicals’ and ‘natural’ have any idea what they are really talking about?
BTW: in my research on this subject, I found out that just because a vanilla ice cream has flecks in it, that does not mean it used vanilla bean vanilla. In fact, the husks of vanilla beans can be bought separately, ground up, and added to just about anything. Presence of black flecks does not indicate ‘all natural’ vanillin.
